Exporting prices: Drug makers’ trade group makes the industry’s priorities US trade policy
Center for Public Integrity, Washington DC
July 7, 2005
Exporting Prices
Drug makers’ trade group makes the industry’s priorities U.S. trade policy
By M. Asif Ismail
WASHINGTON, July 1, 2005 - The Pharmaceutical Research and Manufacturers of America, which represents 67 parent companies and subsidiaries that develop and make prescription and over-the-counter drugs, listed contacts with the government’s top officials overseeing international trade agreements on more lobbying reports than it did for contacts with the Food and Drug Administration, which directly oversees the industry’s products.
PhRMA disclosed contacting the Office of the U.S. Trade Representative on 59 reports filed with the Senate Office of Public Records beginning in 1998, more than any other lobbying organization, the Center for Public Integrity has found.
And USTR, the cabinet-level office which develops and coordinates the country’s international trade policy and oversees negotiations on bilateral and multilateral trade agreements, has incorporated many of the concerns of PhRMA and the companies it represents into official U.S. trade policy.
Intellectual property protections and removing price controls on pharmaceuticals imposed by foreign governments have topped the industry’s agenda. Its heavy lobbying appears to have paid off.
Recent trade negotiations, such as the proposed Dominican Republic-Central American Free Trade Agreement involving the United States, Costa Rica, El Salvador, Guatemala, Honduras, Nicaragua and the Dominican Republic, have incorporated the pharmaceutical companies’ primary demands.
For example, a CAFTA-Dominican Republic draft document stipulates that "export and import price requirements" are prohibited "except as permitted in enforcement of countervailing and antidumping duty orders and undertakings." In other words, under the agreement, countries would be barred from capping the price of pharmaceuticals except as a retaliatory measure against another CAFTA signatory’s unfair trading practices.
Though CAFTA has yet to be adopted, the USTR has been enforcing its provisions. Last year, when the Guatemalan legislature passed a law that permitted lower priced generic drugs to be marketed alongside their brand-name counterparts-a practice that would cut into the profits of American pharmaceutical companies-the USTR responded by threatening to keep Guatemala out of CAFTA. The legislature repealed the measure.
Consumer groups accuse the industry of using the USTR to pressure other countries.
"[The pharmaceutical industry] wants to extend their protection in other parts of the world," said Robert Weissman, an associate counsel at the Consumer Project on Technology, a group focusing on intellectual property rights and health care, among other issues.
Weissman said the USTR has started to pressure other countries to remove price controls. Unlike the United States, which has no cap on drug prices, many countries regulate drug prices.
Collectively, those caps can trim billions of dollars from the industry’s bottom line. In response, the pharmaceutical industry has been lobbying the U.S. government to lobby on its behalf.
In addition to PhRMA, the Biotechnology Industry Organization, another industry trade group representing biomedicine firms, reported lobbying USTR on 35 separate documents-ranking it seventh. Pfizer Inc., the world’s largest drug company, disclosed lobbying the agency 32 times; Bristol-Myers Squibb Co., the ninth largest, lobbied the agency 27 times.
Overall, pharmaceutical firms, their trade organizations and the outside lobbyists that represent them disclosed contacting the USTR on 289 lobbying reports.
Watch list
In addition to battling other nations’ price caps, the USTR has made intellectual property rights, a key pharmaceutical industry concern, into a top-priority issue.
The industry appears to also have a big say on the "watch list" the USTR prepares annually to identify countries that violate the intellectual property rights of U.S. companies in its "Special 301 Report." The Trade Act of 1974, as amended in 1994, requires USTR to identify foreign countries that deny "adequate and effective protection of intellectual property rights" or "nondiscriminatory market access opportunities for United States persons [including corporations] that rely on intellectual property protection."
USTR then issues a "Special 301 Watch List." The list divides countries into three levels: the highest, "Priority Foreign Country," is reserved for those that commit the most consistent and egregious violations of intellectual property rights; the middle level, the "Priority Watch List," is comprised of frequent violators; and, the lowest level, the "Watch List," is for lesser offenders, mainly those countries who tend to raise concerns in only limited areas.
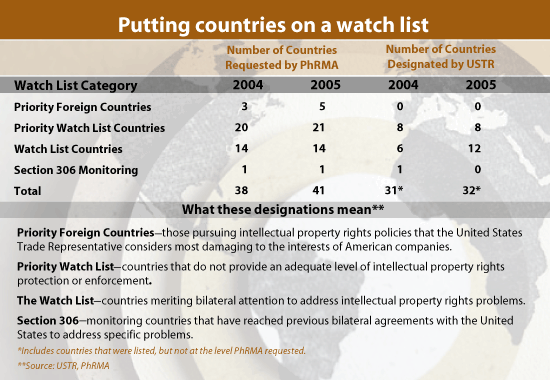
Earlier this year, PhRMA submitted a petition to the USTR recommending 41 countries to be added to varying levels of the agency’s watch list.
Of the 41 countries, USTR placed 20 of them in the 301 report at the level PhRMA suggested. USTR included 12 others cited in PhRMA’s petition in the 301 report, but at lower levels than that suggested by the trade group.
In last year’s petition, PhRMA requested that the USTR take actions against 38 countries for infringing on U.S. patents, producing counterfeit drugs, and releasing confidential test data. USTR placed 15 of them on the list PhRMA requested; 11 of the 15 countries placed on the "Priority Watch List"-the second-highest level for violators-were named specifically by PhRMA.
USTR’s deference to the pharmaceutical industry’s priorities has been noted-and criticized-by some in Congress. "The USTR, in fact, has an office, senior officer for Asia-Pacific and pharmaceutical policy," Rep. Sherrod Brown, D-Ohio, wrote in a recent op-ed. That office, he noted, is "dedicated to assisting the already powerful U.S. drug industry."
The USTR appears to be accomplishing that mission.
Additional reporting by Kevin Boettcher, Alexander Cohen, Victoria Kreha and Emily McNeill





